Gell-Mann proposed the concept of quarks as the fundamental building blocks of protons and neutrons, revolutionizing the understanding of particle physics. Seaborg discovered numerous transuranium elements, expanding the periodic table and contributing to our understanding of the behavior of heavy elements.įeynman made significant contributions to quantum electrodynamics, introducing the famous Feynman diagrams that represent the interactions of subatomic particles. Meitner’s collaborative work on nuclear fission, with Otto Hahn, laid the groundwork for understanding the process that powers nuclear reactors and atomic bombs. This principle remains a fundamental concept in quantum mechanics. Pauli proposed the exclusion principle, stating that no two electrons in an atom can have the same set of quantum numbers. This finding completed the model of the atomic nucleus, providing a more comprehensive understanding of atomic structure.Ĭurie’s pioneering work on radioactivity, along with her discovery of radium and polonium, contributed to the understanding of atomic decay and the existence of subatomic particles.įermi led the team that achieved the first controlled nuclear reaction, opening the door to the development of nuclear power and the atomic bomb. James Chadwick discovered the neutron, a neutral subatomic particle. Maxwell’s equations in electromagnetism laid the groundwork for understanding the relationship between light and electromagnetic waves, contributing to the wave-particle duality concept. Sommerfeld extended Bohr’s model by introducing elliptical orbits and incorporating the concept of azimuthal quantum numbers, refining the description of electron behavior. Pauling’s work on the nature of chemical bonds, including the concept of hybridization, significantly advanced our understanding of molecular structure. See also 10 Importance of Sewing As a Skill Lewis introduced the concept of electron pairs and is renowned for his Lewis dot structures, which provide a simple method for understanding the bonding in molecules. This idea was foundational in the development of quantum theory. Planck introduced the concept of quantization of energy, stating that energy is emitted or absorbed in discrete units called quanta. This concept suggested that particles could display both wave-like and particle-like characteristics. 
His work, along with Heisenberg’s, contributed to the establishment of quantum mechanics.ĭe Broglie proposed that particles, including electrons, exhibit wave-particle duality. Schrödinger developed wave mechanics, providing a mathematical description of the behavior of electrons as waves. 
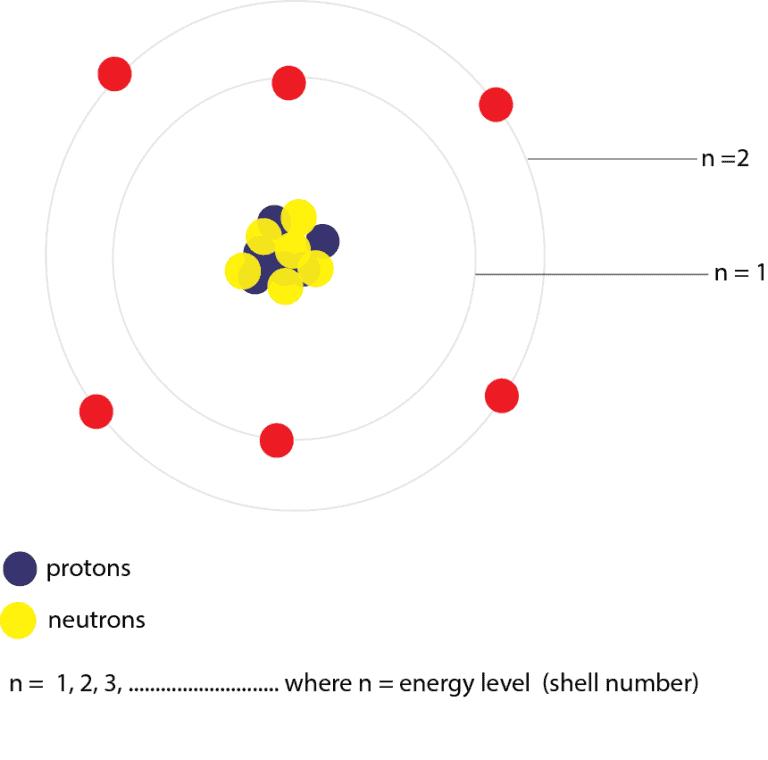
This principle laid the groundwork for quantum mechanics. Heisenberg formulated the uncertainty principle, emphasizing the limits of precision in measuring both the position and momentum of particles. His model explained the spectral lines of hydrogen, and it marked a crucial step towards the development of quantum mechanics. He proposed a nuclear model where electrons orbit a central, positively charged nucleus, drastically altering the understanding of atomic structure.īohr expanded on Rutherford’s work by introducing quantized energy levels for electrons. Rutherford’s gold foil experiment led to the discovery of the atomic nucleus. He proposed the “plum pudding” model, suggesting electrons were embedded in a positively charged matrix. Using cathode ray experiments, Thomson discovered the electron, a subatomic particle with a negative charge. His postulates became the cornerstone for later atomic models. Often hailed as the father of atomic theory, Dalton proposed that matter is composed of indivisible atoms with unique masses. Although his atomic concept lacked empirical evidence, it laid the foundation for future scientific inquiries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
